OFF Research Grant recipient Gracie Liu in the news – 70 per cent of Australian frogs are vulnerable to changes caused by humans
https://www.medianet.com.au/releases/200282/
https://doi.org/10.1111/gcb.15623
In 2017 OFF established a Research Grants fund to assist postgraduate students studying topics relevant to OFF’s mission. The funding comes largely from donations made by OFF members and to date $21,386 has been awarded to 10 postgraduate students. Successful applicants are selected each year by a sub-committee of OFF members and a requirement is that they make a presentation on their research at an OFF meeting.
At the OFF meeting on Monday 26th April 2021 a 2019 grant recipient and UNSW postgrad student Gracie Liu described how Australia’s frogs are coping with human-driven landscape changes and the impacts of chytridiomycosis (the disease that has caused catastrophic declines of hundreds of amphibian species globally). Unfortunately the Zoom recording of Gracie’s talk was poor quality. So instead of loading it on YouTube, we have published a copy of her donor report here.
As well as undertaking her postgraduate research project, Gracie is a member of a UNSW and Australian Museum team using data collected in a citizen science project. They have analysed the distribution of frogs based on frog calls recorded by people all over Australia using the Museum’s FrogID phone app. (Like birds, species of frogs can be identified from their unique calls, but unlike birds, frogs call only during their breeding season which may be just a brief period of the year and usually follows heavy rain).
The FrogID team found 70 per cent of Australian frogs are vulnerable to changes caused by humans, including denser residential development, more intense agriculture and proliferation of roads. The results were published this month and received considerable publicity, including the following quote “Frogs need to be prioritised in urban planning and conservation decisions,” lead author and PhD candidate Gracie Liu from UNSW Science’s School of Biological, Earth and Environmental Sciences says.
Habitat modification and the amphibian chytrid fungus disease
How are multiple threatening processes affecting frogs?
Gracie Liu, The University of New South Wales & The Australian Museum.
Amphibians are one of the most threatened animal groups in the world. Globally, 40% of amphibian species are estimated to be threatened with extinction [1]. In Australia, the numbers are similarly high – almost a fifth of our frog species are threatened [2]. Two of the leading causes of amphibian declines are human-driven habitat modification and disease, and their threats to biodiversity are likely to only increase with the continued expansion of urban and agricultural areas [3, 4].
Chytridiomycosis, a disease caused by the chytrid fungus Batrachochytrium dendrobatidis (Bd), has decimated amphibian populations in recent decades [4]. The disease has caused mass amphibian die-offs and has been implicated in the extinction and decline of hundreds of amphibian species worldwide [4]. While many declines have occurred in undisturbed areas [5], the interaction between disease and habitat modification has the potential to exacerbate species declines. The presence of chytrid is linked to environmental variables such as temperature, moisture, salinity, and canopy cover [6]. At the same time, habitat modification can directly influence environmental characteristics, including temperature and rainfall. However, despite these links, the relationship between habitat modification and disease dynamics is unclear.
Can habitat modification intensify the effects of disease?
To answer this question, I set out to determine chytrid infection prevalence and intensity across a modification gradient. Specifically, I investigated whether infection is related to the degree of habitat modification or species’ habitat use within these different environments. I also examined whether species interactions might be driving patterns of decline.
To test for infection, I swabbed frogs on their bellies, thighs, and feet, using a standard swabbing protocol. I targeted two Australian frog species: the endangered Booroolong frog (Litoria booroolongensis) and the closely related, but stable, stony creek frog (Litoria lesueuri). These species both occur within the NSW Central Tablelands and, in some locations, coexist along the rivers. Given their contrasting threats statuses, comparison of the species pair provided an ideal avenue of examining the subtle drivers of frog declines.

I swabbed frogs across three study locations within the NSW Central Tablelands: Abercrombie River National Park; Fish River; and Essington State Forest. These locations are ~15-68 km apart, and likely represent three distinct populations of frogs. Abercrombie River National Park was gazetted in 1995 and is the most extensive intact area of open forest in the Central Tablelands. The site’s relatively unmodified forests and rivers made it ideal as a reference ‘unmodified’ site. I surveyed five transects within the National Park across all three of the park’s waterways (Retreat River, Abercrombie River and Silent Creek). In contrast, Fish River is surrounded by private and crown land and has been used for recreational and agricultural purposes, including grazing. Essington State Forest is used for forestry operations and, along with Fish River, represents a relatively ‘modified’ site. I established transects along rivers in both these sites where I surveyed and swabbed frogs. These swabs were then taken back to the Australian Museum and processed using quantitative real-time PCR (qPCR) assays to determine infection prevalence and intensity.

I swabbed a total of 163 frogs: 115 Booroolong Frogs and 48 Stony Creek Frogs. The funds provided kindly by the Oatley Flora & Fauna (OFF) Research Fund were instrumental in covering the costs of the project. OFF’s generous donation covered the costs of all the swabs as well as the cost to process more than half of them.

In February 2020, towards the end of the breeding season of both frog species, I swabbed 48 Booroolong frogs and 25 Stony Creek Frogs. Surprisingly, no chytrid was found in any of the 73 frogs. This was a great sign for the frogs in the area, but conclusions could not be drawn about the impacts of habitat modification, so I returned in October 2020 and February 2021 to confirm whether infections levels were indeed very low. I swabbed an additional 90 frogs (67 Booroolong Frogs and 23 Stony Creek Frogs) from across the Central Tablelands, 83 of which have been processed. This time, I detected chytrid, but infection prevalence and intensity were low in both habitats (Figure 1; Table 1). Only eight frogs were infected in total.
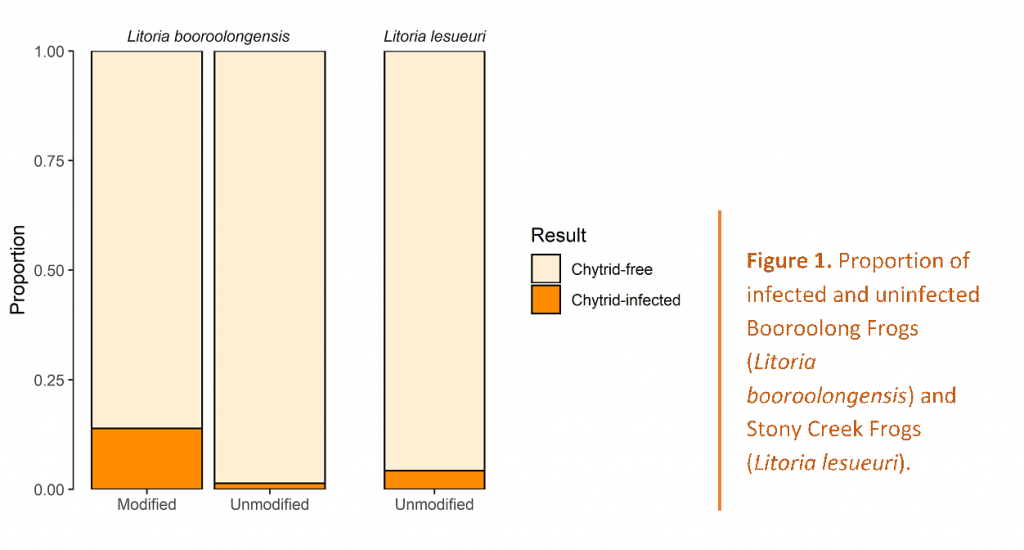
Table 1. Prevalence and intensity (given as zoospore equivalents, ZSE) of chytrid infection in Booroolong Frogs (Litoria booroolongensis) and Stony Creek Frogs (Litoria lesueuri) in unmodified habitat, modified habitat, and in total.
| Number positive | Number sampled | Prevalence (%) | Mean ± s.d. infection intensity (ZSE) (range) | |
| Litoria booroolongensis | ||||
| Unmodified habitat | 1 | 73 | 1.37 | 0 (0–1) |
| Modified habitat | 5 | 36 | 13.9 | 7±38 (0–225) |
| Total | 6 | 109 | 5.50 | 2±22 (0–225) |
| Litoria lesueuri | ||||
| Unmodified habitat | 2 | 47 | 4.26 | 4±30 (0–203) |
Infection prevalence among Booroolong Frogs was less than 2% across the unmodified sites and 14% in the modified sites. The intensity of infection was similarly low. Only one infected individual, which had a zoospore equivalent of one, was detected in the unmodified habitat. Across the modified habitats, the mean zoospore equivalent was seven. These preliminary results suggest that there is some evidence for higher prevalence and intensity of infection among frogs inhabiting modified habitats, but overall, the populations surveyed within the Central Tableland have low rates of infection and infected frogs had low infection loads. This is positive news for these populations of endangered Booroolong Frogs, and with continued conservation management, these sites are likely to be valuable strongholds of this species into the future.
The Stony Creek Frog has previously been identified as a reservoir host species (a species that exhibits high levels of infection, sometimes as high as 78-100%, but persists with the disease and acts as a source of transmission to other frog species) [7-9]. Reservoir species are therefore concerning agents of frog declines [6]. However, in this study system, this does not appear to be the case. Stony Creek Frogs were only found in the unmodified sites, but the prevalence (4%) and intensity of infection (mean zoospore equivalents = 4) were also very low. This is again positive news for both species and suggests that, in this system, Stony Creek Frogs are not significant vectors of chytridiomycosis.
The preliminary results of this project suggest that modified habitats can increase disease transmission, highlighting the importance of considering the impacts of habitat modification and disease in tandem. Past research has suggested that frogs may be better at clearing infection in open habitats compared to closed habitats, but little is known about how frog behaviour or movement may be facilitating this [7]. By combining detailed movement and habitat use data that I have gathered from radiotracking with the results from the current project, I will be able to evaluate whether differences in frog behaviour in modified and unmodified habitats are contributing to disease transmission. The results of this project will form part of my PhD thesis and will be disseminated to a broader audience in the form of a scientific publication or blog. The data from this project, made possible with the support of the Oatley Flora & Fauna Conservation Society, will be important for informing effective conservation management actions for frog species faced with multiple threatening processes.
References
1. Bishop, P., et al., The Amphibian Extinction Crisis-what will it take to put the action into the Amphibian Conservation Action Plan? Surveys and Perspectives Integrating Environment and Society, 2012. 5(2).
2. Gillespie, G.R., et al., Status and priority conservation actions for Australian frog species. Biological Conservation, 2020. 247: p. 108543.
3. Cushman, S.A., Effects of habitat loss and fragmentation on amphibians: a review and prospectus. Biological conservation, 2006. 128(2): p. 231-240.
4. Scheele, B.C., et al., Amphibian fungal panzootic causes catastrophic and ongoing loss of biodiversity. Science, 2019. 363(6434): p. 1459-1463.
5. Saenz, D., T.L. Hall, and M.A. Kwiatkowski, Effects of urbanization on the occurrence of Batrachochytrium dendrobatidis: do urban environments provide refuge from the amphibian chytrid fungus? Urban ecosystems, 2015. 18(1): p. 333-340.
6. Scheele, B.C., et al., After the epidemic: ongoing declines, stabilizations and recoveries in amphibians afflicted by chytridiomycosis. Biological Conservation, 2017. 206: p. 37-46.
7. Van Sluys, M. and J.-M. Hero, How does chytrid infection vary among habitats? The case of Litoria wilcoxii (Anura, Hylidae) in SE Queensland, Australia. EcoHealth, 2009. 6(4): p. 576-583.
8. Brannelly, L.A., et al., Non-declining amphibians can be important reservoir hosts for amphibian chytrid fungus. Animal Conservation, 2018. 21(2): p. 91-101.
9. Crawford-Ash, J. and J.J.L. Rowley, Bad neighbours: amphibian chytrid fungus Batrachochytrium dendrobatidis infection dynamics in three co-occurring frog species of southern Sydney, Australia. Diseases of Aquatic Organisms, 2021. 143: p. 101-108.

